1. Introduction
Compounds are substances made of two or more elements chemically combined.
Compounds are classified into:
- Inorganic (non-carbonic) compounds – obtained from minerals (e.g., salts, oxides).
- Organic (carbonic) compounds – obtained from plants and animals.
- Note: All compounds of carbon except CO, CO₂, carbonates, bicarbonates, and carbides are organic compounds.
- Hydrocarbons are carbonic compounds that contain only carbon (C) and hydrogen (H).
- Examples of hydrocarbons: Methane (CH₄), Ethane (C₂H₆), Propane (C₃H₈).
- Sources of hydrocarbons: Petroleum, natural gas, coal, and plant/animal sources.
2. Organic Compounds
Organic compounds consist mainly of carbon, hydrogen, and sometimes oxygen, nitrogen, halogens, sulfur, or phosphorus.
- They are covalently bonded compounds.
- Examples: Methane, Ethane, Ethene, Acetylene, Methanol, Proteins, Oils.
- Branch of chemistry that studies these compounds: Organic Chemistry or Carbon Chemistry.
Combustibility:
- Combustible substances (e.g., wood, kerosene) → Organic
- Non-combustible substances (e.g., glass, stone) → Inorganic
3. Hydrocarbons
Composed only of carbon and hydrogen.
Main source: Petroleum.
Types based on carbon-carbon bonds:
Saturated hydrocarbons (Alkanes) – single bonds only
Unsaturated hydrocarbons – double or triple bonds
3.1 Saturated Hydrocarbons (Alkanes)
Definition:
Hydrocarbons in which all carbon-carbon bonds are single bonds.
Also called paraffins.
Stable and less reactive compared to unsaturated hydrocarbons.
General formula: CₙH₂ₙ₊₂
Examples of Alkanes
| Name | Molecular Formula | Structural Formula |
|---|---|---|
| Methane | CH₄ | H–C–H |
| Ethane | C₂H₆ | H–C–C–H |
| Propane | C₃H₈ | H–C–C–C–H |
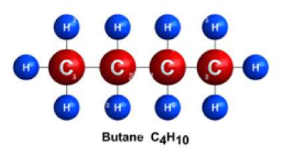
| Butane | C₄H₁₀ | H–C–C–C–C–H |
Key Points:
Saturated → cannot add more hydrogen.
Reactivity: Low → undergo combustion and substitution reactions.
Physical state: Methane, ethane, propane → gases; higher alkanes → liquids/solids.
C–C–C–C–H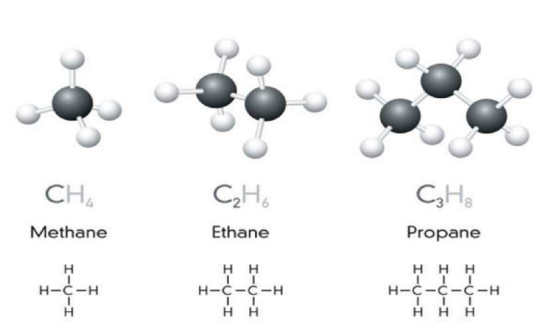
3.2 Unsaturated Hydrocarbons
Definition:
Hydrocarbons that have double or triple bonds between carbon atoms.
Less stable and highly reactive.
Often called olefins.
(a) Alkene – Double Bond Hydrocarbons
General formula: CₙH₂ₙ
Properties: Unsaturated, reactive, undergo addition reactions.
| Name | Molecular Formula | Structural Formula |
|---|---|---|
| Ethene | C₂H₄ | H₂C=CH₂ |
| Propene | C₃H₆ | CH₃–CH=CH₂ |
| Butene | C₄H₈ | CH₃–CH₂–CH=CH₂ |
(b) Alkyne – Triple Bond Hydrocarbons
General formula: CₙH₂ₙ₋₂
Properties: Highly unsaturated, very reactive, undergo addition and combustion reactions.
| Name | Molecular Formula | Structural Formula |
|---|---|---|
| Ethyne | C₂H₂ | HC≡CH |
| Propyne | C₃H₄ | CH₃–C≡CH |
| Butyne | C₄H₆ | CH₃–C≡C–CH₃ |
Key Points:
Alkenes: Double bonds → addition reactions (e.g., hydrogenation).
Alkynes: Triple bonds → more reactive than alkenes.
Both are unsaturated → can form more bonds with hydrogen.
4. Differences between Saturated and Unsaturated Hydrocarbons
| Feature | Saturated (Alkanes) | Unsaturated (Alkene/Alkyne) |
|---|---|---|
| Bond Type | Single bonds only | Double (Alkene) or Triple (Alkyne) bonds |
| Stability | Stable | Less stable, highly reactive |
| General Name | Alkane | Alkene (double bond), Alkyne (triple bond) |
| General Formula | CₙH₂ₙ₊₂ | Alkene: CₙH₂ₙ, Alkyne: CₙH₂ₙ₋₂ |
| Reactivity | Low, undergoes combustion & substitution | High, undergoes addition reactions |
| Hydrogen Content | Maximum hydrogen (saturated) | Can add hydrogen (unsaturated) |
5. Homologous Series
- Definition: Series of hydrocarbons with similar structure, chemical properties, and the same general formula.
- Each member differs by CH₂ unit from the next.
- Example: Methane → Ethane → Propane → Butane
- Molecular weight difference between successive members: 14
6. Alkyl Radical
- Definition: A group formed by removing one hydrogen from an alkane molecule.
- General formula: CₙH₂ₙ₊₁
- Example: –CH₃, –C₂H₅
7. Functional Groups
Definition: Atom or group of atoms that determines the chemical reactivity of an organic compound.
Examples:
- Hydroxyl group: –OH (alcohols)
- Carbonyl group: –CHO (aldehydes)
- Carboxyl group: –COOH (acids)
8. Nomenclature of Hydrocarbons (IUPAC System)
Step 1: Find the root based on number of carbon atoms:
| Number of C atoms | Root |
|---|---|
| 1 | Meth |
| 2 | Eth |
| 3 | Prop |
| 4 | But |
| 5 | Pent |
| 6 | Hex |
| 7 | Hept |
| 8 | Oct |
| 9 | Non |
| 10 | Dec |
Step 2: Add suffix based on bond type:
| Bond Type | Suffix |
|---|---|
| Single | -ane |
| Double | -ene |
| Triple | -yne |
Step 3: Combine Root + Suffix
Example: C₂H₆ → Ethane (2 carbons + single bond → -ane)
Example: C₃H₆ → Propene (3 carbons + double bond → -ene)
Example: C₄H₆ → Butyne (4 carbons + triple bond → -yne)
Key Points:
- Number carbon atoms from the end closest to the double/triple bond.
- For multiple double/triple bonds, use di-, tri-, etc. (e.g., Butadiene, Pentayne).
- Side chains/branches are named as alkyl groups (methyl-, ethyl-, propyl-, etc.) and their position is indicated by number.
9. Important Hydrocarbons and Their Uses
Methane (CH₄)
- Source: Marshy areas, biogas, sewage gas
- Properties: Colorless, odorless, tasteless, insoluble in water
- Uses:
- Cooking fuel (biogas)
- Making carbon black for ink, paint, polish
- Preparation of chloroform, carbon tetrachloride, methanol
Ethane (C₂H₆)
Found in natural gas, petroleum
Uses:
- Heat source in welding
- Making ethyl chloride, nitroethane
Propane (C₃H₈)
- Found in natural gas
- Uses: Fuel, coolant, raw material for carbon compounds
Butane (C₄H₁₀)
- Found in petroleum
- Uses: LPG fuel, synthetic rubber
10. Alcohols
- Compounds derived from alkanes by replacing H with –OH group
- General formula: CₙH₂ₙ₊₁OH
- Functional group: Hydroxyl (–OH)
Types:
- Monohydric (1 –OH) → Methanol, Ethanol
- Dihydric (2 –OH) → Glycol (e.g., Ethane-1,2-diol)
- Trihydric (3 –OH) → Glycerol
Uses
- Methanol: Solvent, formaldehyde, fuels
- Ethanol: Drinks, antiseptics, lab preservative, manufacturing
- Glycol: Coolants, antifreeze
- Glycerol: Soap, cosmetics, sweetening, skin protection
11. Hydrogenation
Process of converting unsaturated fatty acids to saturated fatty acids by adding hydrogen (H₂).
Example: Vegetable oil → Vanaspati ghee
12. Summary
- Hydrocarbons: Only C and H, found in petroleum, natural gas
- Saturated: Single bond, stable, alkanes
- Unsaturated: Double/triple bonds, reactive, alkenes/alkynes
- Organic compounds: Contain C, covalently bonded, derived from plants/animals
- Functional groups define reactivity and type of organic compound
- IUPAC nomenclature gives systematic names