1. Introduction to Heat
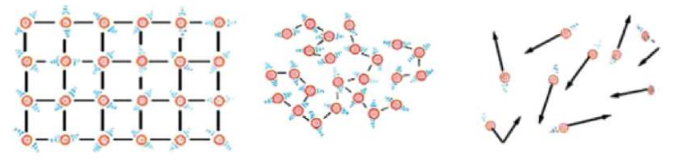
All matter is made up of tiny particles called atoms or molecules. These particles are always in motion.
- Faster motion → higher temperature
- Slower motion → lower temperature
Definition of Heat
Heat is a form of energy transferred from a hotter body to a colder body due to temperature difference.
SI Unit: Joule (J)
Important:
Heat is not stored in a body. What is stored is thermal energy, which is the total kinetic energy of its particles.
2. Thermal Energy
Thermal energy depends on:
- Mass of the substance
- Average kinetic energy of its molecules
Higher temperature → molecules move faster → more kinetic energy.
Example:
A bucket of water at 30°C may have more thermal energy than a cup of boiling water because it contains more molecules.
3. Temperature
Temperature measures average kinetic energy of the particles.
SI unit: Kelvin (K)
Common unit: Celsius (°C)
Important:
0 K = -273.15°C (absolute zero, where molecular motion is theoretically zero).
4. Heat Transfer
Heat is transferred through:
- Conduction
- Convection
- Radiation
(A) Conduction
Transfer of heat through solids without actual movement of particles.
Example:
A metal spoon becomes hot when placed in hot tea.
Reason:
Particles vibrate and transfer energy to neighboring particles.
Metals are good conductors.
Wood and plastic are poor conductors.
(B) Convection
Transfer of heat through liquids and gases by actual movement of particles.
Example:
Warm air rises and cool air sinks forming convection currents.
Applications:
- Sea breeze
- Land breeze
- Boiling water circulation
(C) Radiation
Transfer of heat through electromagnetic waves.
No medium required.
Example:
Heat from the Sun reaches Earth through empty space.
Dark objects absorb more heat.
Light objects reflect more heat.
5. Scientific Reasons Behind Precautions (VERY IMPORTANT)
| Precaution | Method | Scientific Reason |
|---|---|---|
| Do not put hands over boiling water | Convection | Steam carries large thermal energy and causes burns |
| Do not pour hot water into glass in winter | Conduction | Uneven expansion causes glass to crack |
| Do not keep full water bottle in freezer | Expansion | Water expands on freezing and increases pressure |
| Do not touch hot steel plate | Conduction | Metals conduct heat quickly |
6. Effect of Heat on Volume (Expansion)
When heated, particles vibrate faster and move apart → expansion.
Expansion order:
Solids < Liquids < Gases
Solids expand least.
Gases expand most.
7. Iron Ball and Ring Experiment
When heated:
- Molecules move faster
- Ball expands
- Does not pass through ring
When cooled:
- Molecules slow down
- Ball contracts
- Fits through ring again
This proves solids expand on heating.
8. Anomalous Expansion of Water (VERY IMPORTANT)
Water behaves differently from most substances.
Water behaves differently between 0°C and 4°C.
From 0°C to 4°C:
- Water contracts
- Density increases
- Maximum density at 4°C
From 4°C to 0°C:
- Water expands
- Ice becomes less dense
- Ice floats
Effects:
- In ponds and lakes, water at 4°C sinks to the bottom. Water above freezes at the top. Aquatic animals survive below the ice.
- Water pipes burst in winter because water expands on freezing.
- Bottles filled with water crack in freezers.
9. Specific Heat Capacity
Definition:
Specific heat capacity is the heat required to raise the temperature of 1 kg of a substance by 1°C.
Unit: J/kg°C
Heat equation:
Q=msΔT
Where Q = heat gained/lost
m = mass
s = specific heat capacity
ΔT = temperature change
Substances and Their Specific Heat Capacities
- Water = 4200 J/kg°C
Ice = 2100 J/kg°C
Aluminum = 884 J/kg°C
Iron = 460 J/kg°C
Why Water Has High Specific Heat Capacity (Uses)
Because water needs a large amount of heat to increase its temperature:
Uses:
Water is used as a coolant in car radiators and power plants.
- Hot water bags stay warm for long.
- Wet cloth cools a fever patient slowly.
- Coastal areas have smaller temperature differences between day and night.
- Sea breeze and land breeze occur.
10. Sea Breeze and Land Breeze
Sea Breeze (Daytime)
- Land heats faster than sea.
- Warm air above land rises.
- Cool air from sea moves toward land.
Land Breeze (Night)
- Land cools faster than sea.
- Warm air above sea rises.
- Cool air from land moves toward sea.
11. Measurement of Temperature
1. Liquid-in-Glass Thermometer
Contains mercury or alcohol.
When heated, liquid expands and rises.
Scale shows the temperature.
2. Digital Thermometer
Uses a thermistor.
Change in resistance → change in temperature shown on display.
3. Infrared (Radiation) Thermometer
Measures infrared radiation emitted by object.
No contact needed.
12. Calibration of Thermometer
Calibration means marking the scale correctly.
Steps:
- Put bulb in melting ice → mark 0°C (lower fixed point).
- Put bulb in steam from boiling water → mark 100°C (upper fixed point).
- Divide the distance into 100 equal parts → each part = 1°C.
Different scales use different fixed points:
- Celsius: 0°C to 100°C
- Fahrenheit: 32°F to 212°F
- Kelvin: 273 K to 373 K
13. Heat Equation (Summary)
Heat equation: Q = msΔT
Rearranged: s = Q / (mΔT)
Greater mass → more heat needed
Greater temperature difference → more heat needed
COMPLETE SUMMARY FOR QUICK REVISION
- Heat = energy transferred due to temperature difference.
- Temperature = average kinetic energy of particles.
- Thermal energy = total kinetic energy of particles.
- Heat transfer methods: conduction, convection, radiation.
- Expansion: solids < liquids < gases.
- Water shows anomalous expansion.
- Specific heat capacity determines how fast something heats up.
- Water has high SHC → used as coolant.
- Sea breeze (day), land breeze (night).
- Thermometers: liquid-in-glass, digital, radiation.
- Calibration uses fixed points: 0°C and 100°C.
Gallery
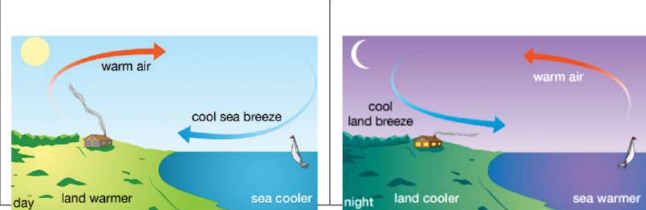
See Breeze
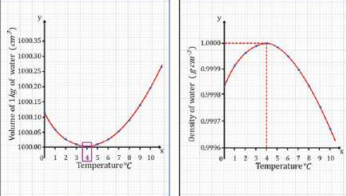
Relation between the volume and density of water, and between the density and temperature of the water