Introduction to Gases
- Suffocation in wells or breathlessness in caves is due to lack of oxygen and presence of certain gases.
- Gases like carbon dioxide (CO₂) and ammonia (NH₃) play significant roles in these phenomena.
- Refrigerators use ammonia gas because of its high solubility, cooling property, and low boiling point.
2. Carbon Dioxide (CO₂)
Sources of Carbon Dioxide
- Natural: Respiration of plants and animals, volcanic eruptions, decomposition of organic matter.
- Artificial: Deforestation, burning of fossil fuels (coal, wood, kerosene, oil, wax).
Discovery:
- Van Helmont (1630) discovered CO₂ by burning wood.
- Joseph Black (1755) prepared CO₂ by heating magnesium carbonate.
- Lavoisier proved it as a compound of carbon and oxygen.
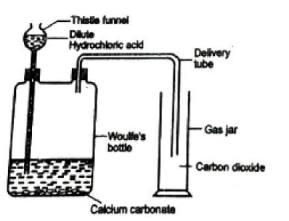
Laboratory Preparation of CO₂
Reaction:
CaCO3+2HCl→CaCl2+H2O+CO2
Apparatus & Chemicals:
- Woulfe’s bottle, gas jar, delivery tube, thistle funnel, rubber cork, limestone/marble powder, dilute HCl, lime water, phenolphthalein, blue litmus.
Procedure:
- Place limestone in Woulfe’s bottle.
- Pour dilute HCl through thistle funnel.
- Collect CO₂ in a gas jar by upward displacement of air.
Tests for CO₂:
- Extinguishes a burning matchstick.
- Turns moist blue litmus red.
- Lime water turns milky due to CaCO₃ formation:
Ca(OH)2+CO2→CaCO3+H2O
- Prolonged CO₂ passage makes it colorless due to Ca(HCO₃)₂ formation:
CaCO3+CO2+H2O→Ca(HCO3)2
Properties of CO₂
Physical:
- Colourless, odourless, slightly soluble in water, heavier than air (1.5×).
- Acidic, non-toxic but causes suffocation in high concentration.
- Forms dry ice at -78°C.
Chemical:
- Non-combustible and does not support combustion.
- Burns magnesium to form MgO and carbon:
2Mg+CO2→2MgO+C2
- Forms carbonic acid in water:
CO2+H2O→H2CO3
- Used in soft drinks, fire extinguishers, dry ice, sugar purification, urea, baking soda, carbogen, photosynthesis.
3. Ammonia (NH₃)
Sources of Ammonia
- Free state: Produced by decay of nitrogenous substances in absence of oxygen.
- Combined form: Found in ammonium chloride (NH₄Cl) and ammonium sulfate [(NH₄)₂SO₄].
Laboratory Preparation:
2NH4Cl+Ca(OH)2→CaCl2+2H2O+2NH3
Apparatus & Chemicals:
Hard glass test tube, lime tower, delivery tube, stand, ammonium chloride, calcium hydroxide.
Procedure:
Mix NH₄Cl and Ca(OH)₂ in a 2:1 ratio in test tube.
Heat gently, pass gas through lime tower to get pure dry ammonia.
Tests for NH₃:
Turns red litmus blue (basic gas).
Produces white fumes with concentrated HCl.
Properties of Ammonia
Physical:
Colourless, pungent odour, lighter than air, highly soluble in water, liquefies at -33.4°C, solidifies at -78°C.
Chemical:
Forms ammonium hydroxide in water:
NH3+H2O→NH4OH
Reacts with acids to form ammonium salts:
NH4OH+HCl→NH4Cl+H2O
Reacts with CO₂ at 1500°C under high pressure to form urea:
2NH3+CO2→NH2−CO−NH2+H2O
Burns in oxygen to form nitrogen and water.

Uses of Ammonia
- Fertilizers: urea, ammonium sulfate, ammonium nitrate.
- Manufacturing: nitric acid, washing soda, plastics, medicines.
- Refrigerants, cleansing agent, blueprint production.
- Activity: Construction of ammonia fountain demonstrates its high solubility and basic nature.
4. Greenhouse Effect
- Natural Greenhouse: Earth’s atmosphere traps solar radiation through gases like CO₂, CH₄, N₂O, O₃, maintaining a habitable temperature.
- Artificial Greenhouse: Transparent plastic/glass structures trap solar heat, useful for growing off-season plants.
- Mechanism:
- Short-wave radiation enters → absorbed by surfaces → converted to long-wave radiation → trapped → raises temperature.
Consequences of Enhanced Greenhouse Effect
- Global temperature rise, climate change.
- Melting glaciers, rising sea levels, flooding.
- Loss of biodiversity, desertification, ecosystem imbalance.
- Health impacts and decreased agricultural productivity.
Ways to Reduce Greenhouse Effect
- Reduce CFCs and fossil fuel usage.
- Promote renewable energy: solar, wind, hydroelectricity.
- Plant more trees.
- Reduce industrial CO₂ emissions.
5. Acid Rain
- Formed when gases like SO₂, CO₂, NOx, Cl₂ mix with water vapor to produce acids: H₂SO₄, H₂CO₃, HNO₃, HCl.
- pH of acid rain: 3–5 (normal rain ≈ 6).
Chemical Reactions:
- SO2+H2O→H2SO3
- CO2+H2O→H2CO3
Effects of Acid Rain
- Erodes marble monuments and buildings.
- Increases soil acidity → reduces crop productivity.
- Pollutes water bodies → harms aquatic life.
- Causes skin diseases and health issues.
Prevention
- Reduce SO₂ and NOx emissions.
- Promote renewable energy.
- Raise awareness about acid rain.
Summary Table
| Gas | Symbol | Properties | Uses |
|---|---|---|---|
| Carbon dioxide | CO₂ | Colourless, odourless, heavier than air, acidic | Soft drinks, dry ice, fire extinguishers, sugar purification, photosynthesis |
| Ammonia | NH₃ | Colourless, pungent, lighter than air, basic, highly soluble | Fertilizers, refrigerants, plastics, cleaning agent, urea |