Classification of Elements
1. Introduction
- Matter around us exists as elements, compounds, and mixtures.
- There are 118 elements, each with unique properties.
- To study them systematically, scientists classified elements based on similarities in physical and chemical properties.
Definition of Classification of Elements:
Arrangement of elements in a systematic manner according to their similar properties.
Purpose of Classification:
- Elements have different physical and chemical properties.
- Some elements show similar properties.
- Helps in studying, predicting, and understanding the behavior of elements easily.
2. Historical Developments
(a) Döbereiner’s Triads (1829)
- Elements grouped in triads of three with similar properties.
- Atomic mass of the middle element ≈ average of the other two.
- Example: Li (7), Na (23), K (39) → 23 ≈ (7 + 39)/2
- Limitation: Worked only for a few triads.
(b) Newlands’ Law of Octaves (1864)
- Arranged elements by increasing atomic mass.
- Every 8th element had similar properties to the first.
Limitations:
- Worked only up to calcium.
- Failed for heavier elements.
- Transition metals did not fit.
(c) Lothar Meyer’s Curve (1869)
- Plotted atomic volume vs atomic mass.
- Peaks corresponded to alkali metals.
- Showed periodicity of properties.
(d) Mendeleev’s Periodic Table (1869)
Arranged elements by increasing atomic mass.
Achievements:
- Left gaps for undiscovered elements.
- Predicted properties of Ga, Ge, Sc accurately.
- Grouped elements correctly.
Limitations:
- Atomic mass order sometimes failed (Co & Ni).
- Position of hydrogen unclear.
- Isotopes did not fit.
3. Modern Periodic Law
"Properties of elements are a periodic function of their atomic number (Z)."
Atomic number is the real basis of periodicity.
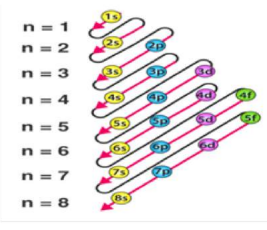
4. Modern Periodic Table Features
Periods (Horizontal Rows): 7
Period number = number of electron shells (n)
Groups (Vertical Columns): 18
Group number = number of valence electrons (except transition metals)
Blocks:
- s-block: Groups 1–2 → Highly reactive metals, form basic oxides
- p-block: Groups 13–18 → Metals, non-metals, metalloids, halogens, noble gases
- d-block: Groups 3–12 → Transition metals, colored compounds, variable oxidation states
- f-block: Lanthanides & Actinides → Inner transition elements, mostly radioactive (actinides)
5. Key Differences
Metals vs Non-metals
| Metals | Non-metals |
|---|---|
| Shiny | Dull |
| Good conductors | Poor conductors |
| Form cations | Form anions |
| Malleable | Brittle |
s-block vs p-block
| s-block | p-block |
|---|---|
| Groups 1–2 | Groups 13–18 |
| Highly reactive | Mixed types |
| Simple electron config | Complex behavior |
6. Periodic Properties
(A) Atomic Radius
Across a period: Decreases (more nuclear charge pulls electrons closer)
Down a group: Increases (more electron shells added)
(B) Ionic Radius
Cations (+): Smaller (lose electrons)
Anions (−): Larger (gain electrons)
(C) Ionization Energy (IE)
- Energy required to remove an electron.
- Across period → increases
- Down group → decreases
- Low IE → Alkali metals; High IE → Noble gases
(D) Electron Affinity (EA)
- Energy released when an atom gains an electron.
- Across period → increases
- Down group → decreases
- Highest EA → Chlorine
(E) Electronegativity (EN)
- Tendency to attract shared electrons.
- Across period → increases
- Down group → decreases
- Most EN element → Fluorine
(F) Metallic Character
- Decreases from left → right
- Increases from top → bottom
- Opposite trend to non-metallic character
7. Special Cases
- Hydrogen: Behaves like alkali metals and halogens → special position
- Helium: Noble gas, though s-block
- Lanthanide contraction: Affects d-block sizes
- Co & Ni anomaly: Fixed in modern table
8. Valency
- Valency: Number of electrons an atom gains, loses, or shares.
- s-block: Valency = group number
- p-block: Varies (commonly 3, 4, 5, etc.)
9. Why Periodicity Occurs?
Because electronic configurations repeat at regular intervals.
Example:
Group 1: ns¹
Group 2: ns²
Group 17: ns² np⁵
Gallery
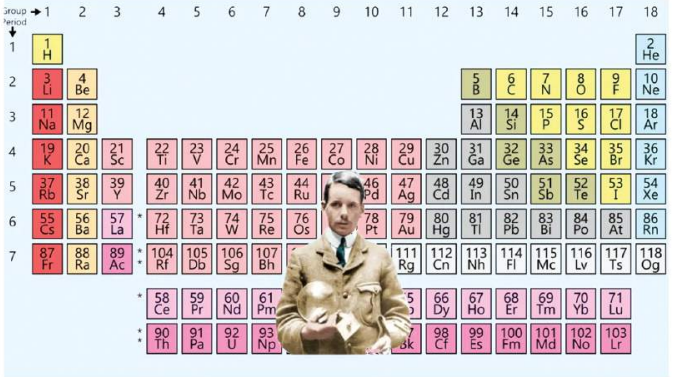
Henry Moseley and Periodic Table

Modern periodic Table