1. Meaning of Scientific Study
Scientific study is a systematic process of gaining knowledge about natural phenomena based on observation, experiment, data collection, and logical reasoning.
Science is not guesswork; it is based on facts, evidence, and experiments.
2. Steps of Scientific Learning (Scientific Method)
The steps of scientific learning are shown in Fig. 1.2.
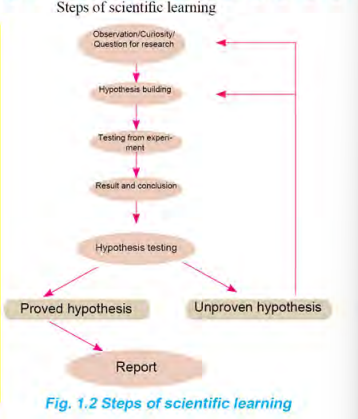
🔬 Steps:
Observation / Curiosity
Question for research
Hypothesis building
Experiment / Testing
Result and conclusion
Hypothesis testing
Report and communication
Explanation:
Observation: Careful watching of events (e.g., sour taste of lemon)
Question: Why is lemon sour?
Hypothesis: Lemon is acidic.
Experiment: Test lemon juice using pH paper.
Data collection: pH value recorded.
Conclusion: Lemon is acidic.
Hypothesis test:
✔ Proved hypothesis
✘ Unproven hypothesis (needs modification)
3. Scientific Study of Acidic, Basic and Neutral Substances

(a) Research Question
- Are fruits and chemicals acidic, basic, or neutral?
(b) Hypothesis
- Lemon and orange are acidic.
- Soap and washing powder are basic.
- Water and salt solution are neutral.
(c) Experimental Work
Test substances using pH paper / universal indicator
Observe color change
(d) Data Collection Method
Record pH value
Compare with pH scale:
pH < 7 → Acidic
pH = 7 → Neutral
pH > 7 → Basic
4. Scientific Process Skills
Scientific process skills help us learn science properly.
Important skills:
- Observation
- Questioning
- Classification
- Prediction
- Measurement
- Interpretation
- Conclusion
Communication
5. Activity: Paper Aeroplane

Scientific skills used:
- Observation
- Prediction
- Experimentation
- Comparison
- Conclusion
Example:
Predict which aeroplane flies longer → test → compare → improve model.
6. Scientific Learning from Daily Life
Example: Formation of water drops

Cold plate → more water drops
Hot plate → fewer water drops
Conclusion:
Temperature affects condensation.
Same principle is used in:
Cloud formation
Rainfall prediction
Weather forecasting
7. Scientific Study in Vaccine Development

Steps followed:
Observation of virus effect
Question: How to stop virus?
Hypothesis: Vaccine can protect
Testing on animals (mice)
Data collection
Testing on humans
Mass application
This shows real-life application of scientific method.
8. Fields of Science
(A) Biology
Study of living organisms
Branches: Zoology, Botany, Genetics, Microbiology
Professionals: Doctor, Biologist, Genetic Engineer
(B) Physics
Study of force, motion, energy, matter
Branches: Nuclear physics, Atomic physics
Professionals: Engineer, Physicist, Radiologist
(C) Chemistry
Study of matter and chemical changes
Branches: Organic, Inorganic, Physical chemistry
Professionals: Chemist, Pharmacologist
(D) Interdisciplinary Fields
Geology, Astronomy, Environmental Science, Agriculture
9. Achievements of Science and Technology
| Field | Achievements |
|---|---|
| Biology | Vaccine, organ transplant, cloning |
| Physics | X-ray, microscope, communication |
| Chemistry | Medicine, fertilizer, plastics |
| Agriculture | Improved seeds, higher yield |
| AI | Automation, smart technology |
10. Challenges of Science and Technology
- Radiation hazards
- Air and noise pollution
- Environmental degradation
- Health problems
- Privacy issues in AI
- Unemployment due to automation
11. Safety Measures in Scientific Experiments
Important rules:
- Perform experiments under teacher supervision
- Wear lab coat, gloves, safety goggles
- Never add water to acid (add acid to water)
- Heat alcohol using water bath
- Use chemicals carefully
- Inform teacher in case of accident
- Clean lab after experiment
- Example: Marie Curie died due to radiation exposure.
12. Scientific Measurement
Measurement is essential in science to get accurate data.
Types of measurement:
Small (virus size)
Large (distance between Earth and Sun)
13. Scientific Notation
Definition:
A method of expressing very large or very small numbers in powers of 10.
Rules:
Base is always 10
Coefficient is between 1 and 9
Power is positive or negative
Examples:
1230000 = 1.23 × 10⁶
0.00042 = 4.2 × 10⁻⁴
0.00000001 = 1 × 10⁻⁸
300000000 = 3 × 10⁸
14. Metric Prefix
| Prefix | Symbol | Value |
|---|---|---|
| kilo | k | 10³ |
| mega | M | 10⁶ |
| giga | G | 10⁹ |
| milli | m | 10⁻³ |
| micro | μ | 10⁻⁶ |
| nano | n | 10⁻⁹ |
1 nanosecond = 10⁻⁹ second
15. Least Count of Instruments
Definition:
The smallest measurement an instrument can measure.
| Instrument | Quantity Measured | Least Count |
|---|---|---|
| Scale | Length | 1 mm |
| Stopwatch | Time | 0.01 s |
| Protractor | Angle | 1° |
Smaller least count = more accurate measurement
16. Importance of Average in Measurement
Measurements may vary
Taking average reduces error
Average = (Sum of readings) / Number of readings
Average measurement is more reliable and accurate.